COVID-19 Ag Card Test
The BinaxNow™ COVID-19 Ag Test is FDA Approved. Get Fast, Accurate Results in less than 15 minutes. It is intended to aid in the rapid diagnosis of SARS-CoV-2 infections. The Coronavirus Ag Antigen Test Kit (Swab) does not differentiate between SARS-CoV and SARS-CoV-2. As an intended Point-of-Care (POC) designated test with a 15 minute read processing time.
Features
- Method: Lateral Flow
- Detect SARS-CoV-2 nucleocapsid protein antigen
- Rapid results are read at 15 minutes
- Intended at POC setting (i.e., in patient care settings) by medical professionals operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation
- For use under the Emergency Use Authorization (EUA) only
- For prescription use only
- For use with direct anterior nasal and nasopharyngeal swab specimens
Clinical Features
- Detect SARS-CoV-2 nucleocapsid protein antigen
- 40 Patient COVID-19 Fact Sheets
- Sensitivity (PPA) 84.6%
- Specificity (NPA) 95.6%
aPPA: Positive Percent Agreement, bNPA: Negative Percent Agreement.
COVID-19 Antigen Test Components:
- 40 Test Cards
- 40 Sterile Nasal Swabs
- Buffers
- Package Insert
- Instruction for Use
- Healthcare Provider COVID-19 Fact Sheet
Procedure & Preparation
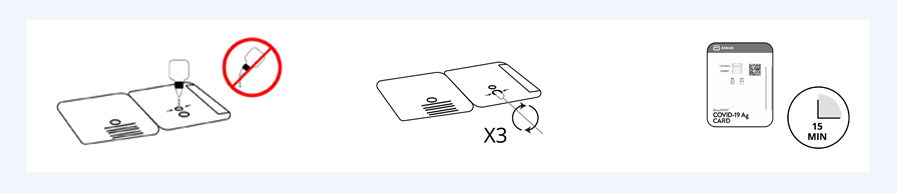
Specimen Collection
- Open the cassette test card and put three drops of the reagent in the below hole.
- Remove the swab by rotating against the extraction vial while rotating the nasal swap shaft of the vial to collect the liquid to the swab.
- Insert the swab by rotating against the above hole of the cassette test card while rotating the nasal swap shaft three times of the hole to release the liquid from the swab. Properly discard the swab.
- Close the cassette test card and wait for 15 minutes for result.
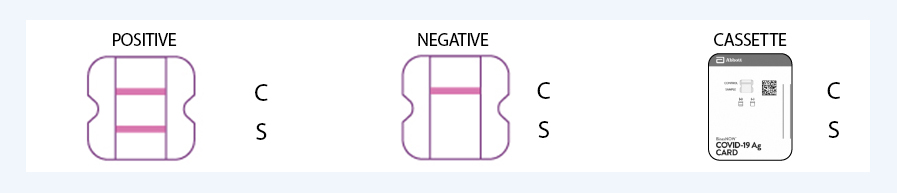
Covid-19 Antigen Rapid Test Kit Results Interpretation
Sample Results Interpretation
Fast result at 15 minutes. The test result should not be read after 15 minutes
|
Positive
|
Negative
|
Read Result
|
|
SARS-CoV-2 antigen present does not rule out co-infection with other pathogens. The color intensity in the test region will vary depending on the amount of SARS-CoV-2 antigen present in the sample. Any faint colored line(s) in the test region(s) should be considered as positive.
|
Negative test results do not preclude infection and should not be used as the sole basis for treatment or other patient management decisions, including infection control decisions. It is recommended that these results be confirmed by a molecular testing method, if necessary for patient management.
|
Re-run the test one time using the remaining specimen in the extraction vial if an invalid result is obtained during initial testing.
|
Warning: The false positive, false negative, or invalid results may occur if the test is interpreted outside of the interpretation window.
‾
Before testing patients, federal regulations require testing sites to have a CLIA certificate issued by CMS. Sites performing only waived tests must obtain a Certificate of Waiver by applying for this certification for each location performing testing. This product has not been FDA cleared or approved, but has been authorized by FDA under an EUA for use by authorized laboratories. This product has been authorized only for the detection
of proteins from SARS-CoV-2, not for any other viruses or pathogens; and, The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug, and Cosmetic Act, 21 U.S.C. § 360bbb3(b)(1), unless the declaration is terminated or authorization is revoked sooner.